For the octahedral complexes of Fe3+ in SCN– (thiocyanato - S) and in CN– ligand environments, the difference between the spin only magnetic moments in Bohr magnetons (when approximated to the nearest integer) is:
[Atomic number of Fe = 26]
[Fe(SCN)6]3- and [Fe(CN)6]3-
In both the cases the electronic configuration of Fe3+ will be
1s2, 2s2, 2p6, 3s2, 3p6, 3d5
Since SCN is a weak field ligand and is a strong field ligand, the pairing will occur only in case of [Fe(CN)6]3-
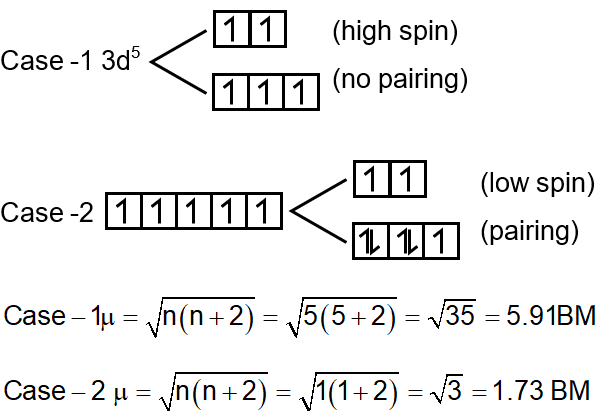
Difference in spin only magnetic moment = 5.91 – 1.73 = 4.18 ≈ 4
This question involves calculating the spin-only magnetic moments for Fe3+ complexes with different ligands (SCN– and CN–) and finding the difference between them. The magnetic moment depends on the number of unpaired electrons, which is determined by whether the complex is high-spin or low-spin. This, in turn, depends on the strength of the ligand field.
Atomic number of Fe is 26. Its electron configuration is [Ar] 4s2 3d6.
For Fe3+, three electrons are removed. Typically, electrons are removed from the 4s orbital first, followed by the 3d orbital.
Therefore, the configuration of Fe3+ is [Ar] 3d5.
This means there are 5 electrons in the d orbitals.
In an octahedral field, the five d orbitals split into two sets: the lower-energy t2g set and the higher-energy eg set. The energy difference between them is called Δo (crystal field splitting energy).
Whether a complex is high-spin (maximum unpaired electrons) or low-spin (minimum unpaired electrons) depends on the strength of the ligand:
From the spectrochemical series:
SCN– (bonding through S) is a weak field ligand.
CN– is a strong field ligand.
Therefore:
For a d5 system:
High-spin complex (weak field, e.g., SCN–): Electrons will remain unpaired to maximize spin. All five electrons are unpaired.
Number of unpaired electrons, = 5.
Low-spin complex (strong field, e.g., CN–): Electrons will pair up. The t2g set can hold 6 electrons. All 5 electrons will pair up as much as possible, leaving only one unpaired electron.
Number of unpaired electrons, = 1.
The spin-only magnetic moment () is given by the formula:
Where is the number of unpaired electrons and BM stands for Bohr Magnetons.
For [Fe(SCN)6]3– (High-spin, n=5):
Approximated to the nearest integer: BM.
For [Fe(CN)6]3– (Low-spin, n=1):
Approximated to the nearest integer: BM.
Difference = |μhigh - μlow| = |6 - 2| = BM.
The difference between the spin-only magnetic moments is Bohr Magnetons.
Key Topics:
Important Formulae:
Spin-only magnetic moment:
Theory: The spin-only formula is derived from the quantum mechanical treatment of electron spin. It provides a good approximation for the magnetic moment of transition metal complexes, especially for 3d metals where orbital contributions are often quenched. The actual magnetic moment can be slightly higher than the spin-only value if there is a contribution from the orbital angular momentum.