The % yield of ammonia as a function of time in the reaction
N2(g) + 3H2(g) ⇌ 2NH3 (g), ΔH < 0
at (P, T1) is given below:
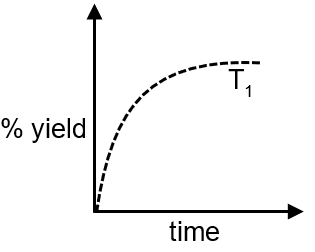
If this reaction is conducted at (P, T2) , with T2 > T1, the % yield of ammonia as a function of time is represented by :
N2(g) + 3H2(g)
N2(g) + 3H2(g) 2NH3(g) ; H < 0
The reaction is:
This is an exothermic reaction (ΔH < 0), meaning it releases heat. According to Le Chatelier's Principle, increasing temperature favors the endothermic direction. Since the forward reaction is exothermic, increasing temperature will shift the equilibrium toward the reactants, reducing the yield of ammonia.
Step 1: At temperature T1, the % yield of ammonia increases with time until equilibrium is reached, as shown in the given graph.
Step 2: When temperature is increased to T2 (T2 > T1), the equilibrium shifts left (reverse reaction favored), reducing the maximum possible % yield of ammonia at equilibrium.
Step 3: However, increasing temperature also increases the rate of reaction (both forward and reverse), so equilibrium is reached faster.
Step 4: Therefore, at T2, the curve will:
Final Answer: The correct graph is the one where the curve rises faster but levels off at a lower % yield compared to the original curve at T1.
Le Chatelier's Principle: When a system at equilibrium is disturbed, it adjusts to minimize the effect of the disturbance. For temperature: increasing temperature favors the endothermic direction.
Effect of Temperature on Equilibrium: For exothermic reactions, increasing temperature decreases the equilibrium constant (K) and product yield. For endothermic reactions, it increases them.
Reaction Kinetics: Higher temperature increases reaction rate due to more frequent and energetic collisions, leading to faster attainment of equilibrium.
Van't Hoff Equation (for temperature dependence of K):
For exothermic reactions (ΔH° < 0), if T2 > T1, then K2 < K1.