Match the chemical conversions in List I with the appropriate reagents in List II and select the correct answer using the code given below the lists:
| List I | List II |
| (1) (i) Hg(OAc)2; (ii) NaBH4 | |
| (2) NaOEt | |
 |
(3) Et – Br |
 |
(4) (i) BH3 ; (ii) H2O2/NaOH |
This question involves matching organic chemical conversions with the correct reagents. Each conversion represents a specific type of organic reaction. Let's analyze each conversion step by step:
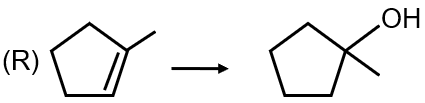
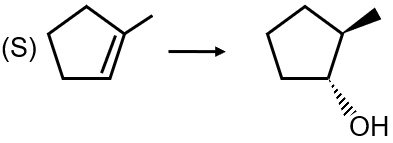
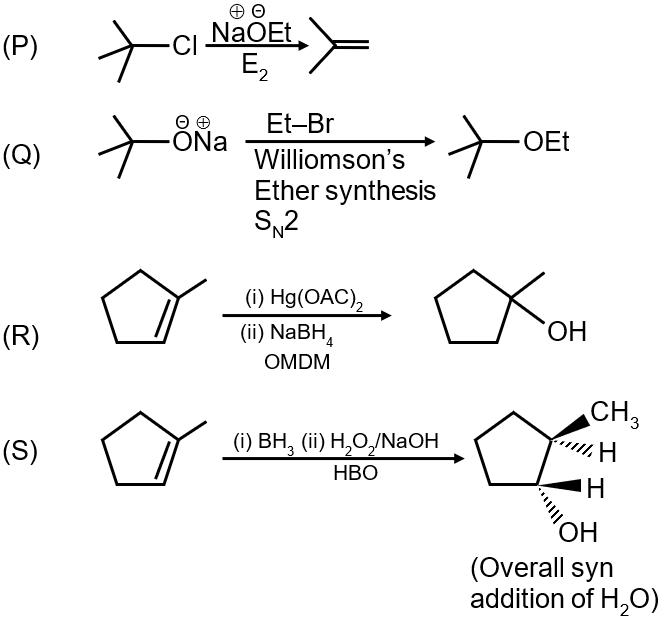
Conversion 1: The reaction shows an alkene being converted to an alcohol with anti-Markovnikov orientation. The reagent that achieves this is (i) BH3 followed by (ii) H2O2/NaOH, which is hydroboration-oxidation. This matches with option (4).
Conversion 2: This shows an intramolecular Williamson ether synthesis where an alkoxide attacks an alkyl halide to form a cyclic ether. The base NaOEt (sodium ethoxide) is used to deprotonate the alcohol and generate the alkoxide nucleophile. This matches with option (2).
Conversion 3: This shows an alkene undergoing oxymercuration-demercuration to form an alcohol with Markovnikov orientation. The reagents are (i) Hg(OAc)2 followed by (ii) NaBH4. This matches with option (1).
Conversion 4: This shows a carboxylic acid being converted to its ester through reaction with an alkyl halide (Et-Br) in the presence of base, which is a common method for esterification. This matches with option (3).
Therefore, the correct matching is: 1→4, 2→2, 3→1, 4→3.
Alkene Reactions: Alkenes undergo various addition reactions including hydroboration-oxidation (anti-Markovnikov addition) and oxymercuration-demercuration (Markovnikov addition). Understanding the mechanism and stereochemistry is crucial.
Ether Formation: Williamson ether synthesis involves SN2 reaction between an alkoxide ion and an alkyl halide. Intramolecular versions form cyclic ethers.
Esterification: Carboxylic acids can be converted to esters using various methods including reaction with alkyl halides in the presence of base.
Hydroboration-Oxidation: BH3·THF followed by H2O2/NaOH gives anti-Markovnikov alcohol with syn addition.
Oxymercuration-Demercuration: Hg(OAc)2/H2O followed by NaBH4 gives Markovnikov alcohol without rearrangement.
Williamson Ether Synthesis: R-ONa + R'-X → R-O-R' + NaX (SN2 mechanism)
Esterification: RCOOH + R'X → RCOOR' + HX (with base to deprotonate acid)