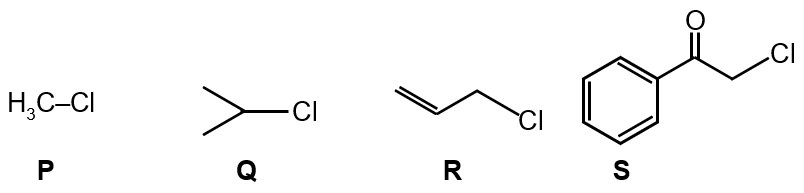
KI in acetone, undergoes SN2 reaction with each of P, Q, R and S. The rates of the reaction vary as

Rate of SN2
+ charge on carbon
Stability of transition state
So, S > R > P > Q
In SN2 reactions, the rate depends on steric hindrance and the leaving group ability. For alkyl halides reacting with KI in acetone (a polar aprotic solvent that favors SN2):
Step 1: Identify the carbon center and its substitution
P: CH₃-CH₂-Br (primary alkyl bromide)
Q: (CH₃)₂CH-Br (secondary alkyl bromide)
R: CH₂=CH-CH₂-Br (allylic bromide, primary but with resonance stabilization)
S: Ph-CH₂-Br (benzylic bromide, primary with resonance stabilization)
Step 2: Compare steric hindrance
Primary halides (P, R, S) have less steric hindrance than secondary (Q). So Q is slowest.
Step 3: Consider resonance effects
Allylic (R) and benzylic (S) halides have resonance stabilization of the transition state, making them react faster than simple primary halides (P). Between R and S, benzylic (S) has better resonance stabilization than allylic (R) due to the aromatic ring.
Step 4: Order the rates
Benzylic (S) > Allylic (R) > Simple primary (P) > Secondary (Q)
So the order is: S > R > P > Q
Final Answer: The correct option is S > R > P > Q
The rate law for SN2 reaction is:
General reactivity order for SN2: CH₃X > RCH₂X (primary) > R₂CHX (secondary) > R₃CX (tertiary, very slow)
Special cases: Benzylic and allylic halides undergo SN2 faster than simple alkyl halides due to resonance stabilization of the transition state.
Carbocation stability order (for SN1): tertiary > secondary > primary > methyl, and benzylic ≈ allylic > tertiary.