Given :
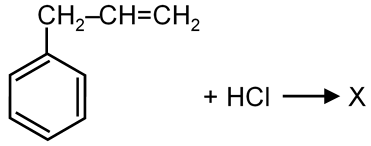
X is :
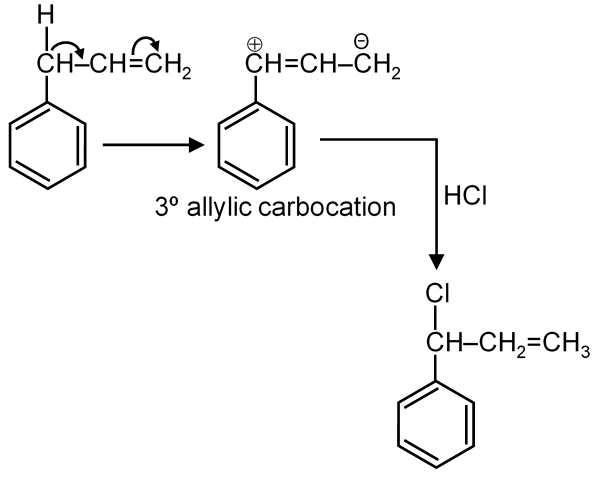
(Since tertiary carbocation is more stable)
Given the reaction:
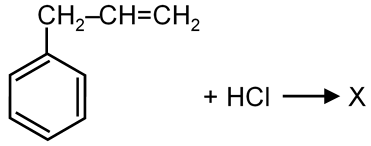
We need to identify the product X. This is a classic example of the Wurtz Reaction.
The reaction shows an alkyl halide (specifically, an alkyl bromide) reacting with sodium metal in dry ether. This is the condition for the Wurtz reaction.
The general reaction is:
Where two molecules of an alkyl halide couple to form a new carbon-carbon bond, producing a symmetrical alkane with twice the number of carbon atoms.
The given reactant is Bromoethane (). It is a primary alkyl halide with 2 carbon atoms.
When Bromoethane undergoes the Wurtz reaction, two molecules will combine.
The product is a symmetrical alkane: Butane ().
We need to find the structure of Butane among the given options.
Butane has the condensed formula CH₃-CH₂-CH₂-CH₃. Its structure is a straight chain of four carbon atoms.
Comparing the options:
Therefore, the correct option is the second one.
X is Butane.
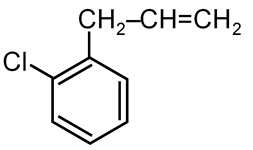
Theory: The Wurtz reaction is a coupling reaction in organic chemistry where two alkyl halides react with sodium metal in dry ether solution to form a higher alkane. It is particularly useful for preparing symmetrical alkanes with an even number of carbon atoms.
Limitations:
The general chemical equation for the Wurtz Reaction:
Where X = Cl, Br, I