In the following sequence of reactions :
Toluene the product C is :
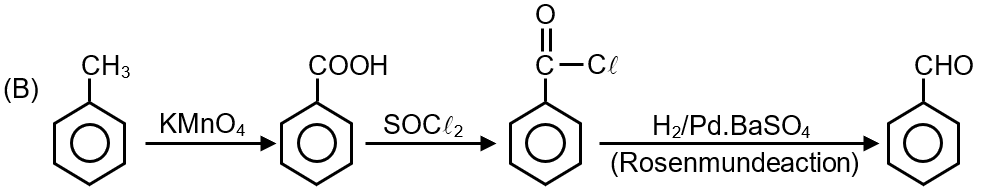
This question involves a sequence of organic reactions starting from toluene. Let's analyze each step to identify the final product C.
Toluene (C6H5CH3) undergoes oxidation with potassium permanganate (KMnO₄). The alkyl side chain (methyl group) attached to the benzene ring is oxidized to a carboxylic acid group, regardless of the chain length (as long as it has a benzylic hydrogen).
Reaction:
So, product A is Benzoic acid (C6H5COOH).
Benzoic acid (A) reacts with thionyl chloride (SOCl₂). This reaction converts the carboxylic acid group (–COOH) into an acid chloride group (–COCl).
Reaction:
So, product B is Benzoyl chloride (C6H5COCl).
Benzoyl chloride (B) undergoes reduction with hydrogen gas in the presence of a palladium catalyst poisoned with barium sulfate (H2/Pd-BaSO4). This specific reaction is known as the Rosenmund reduction. It selectively reduces acid chlorides to aldehydes.
Reaction:
So, the final product C is Benzaldehyde (C6H5CHO).
Therefore, the product C is C6H5CHO (Benzaldehyde).
Oxidation of Alkylbenzenes: Alkylbenzenes with a benzylic hydrogen are oxidized to benzoic acids by strong oxidizing agents like KMnO₄.
Conversion of Carboxylic Acids to Acid Chlorides: Carboxylic acids react with SOCl₂, PCl₅, or PCl₃ to form acyl chlorides. General reaction: R-COOH + SOCl₂ → R-COCl + SO₂ + HCl
Rosenmund Reduction: A specific reaction where acid chlorides are reduced to aldehydes using hydrogen gas over a palladium catalyst poisoned with barium sulfate. This poisoning is crucial to prevent over-reduction to the primary alcohol. General reaction: R-COCl + H₂ → R-CHO + HCl