How many chiral compounds are possible on monochlorination of 2-methyl butane ?
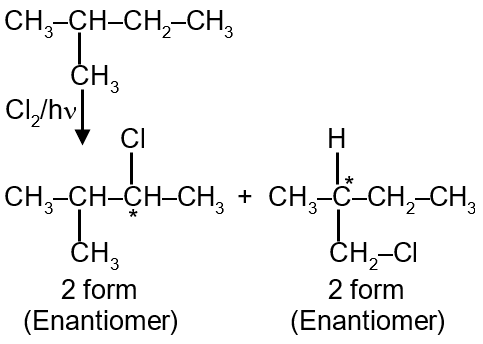
Total 4 Chiral molecules
Chiral compounds are molecules that are non-superimposable on their mirror images, typically due to the presence of a chiral center (a carbon atom with four different substituents). In monochlorination, we replace one hydrogen atom with a chlorine atom. To determine how many chiral compounds form, we must identify all unique monochlorinated products and check which have chiral centers.
2-Methylbutane (C5H12) has the carbon skeleton:
Carbon 1: CH3- | Carbon 2: CH- (with CH3 branch) | Carbon 3: CH2- | Carbon 4: CH3
Written as: CH3-CH(CH3)-CH2-CH3
Different hydrogens yield different products upon substitution. Classify by equivalence:
Total hydrogens: 9 (a) + 2 (b) + 1 (c) = 12, correct for C5H12.
Substituting each type gives:
Only the product from substituting Type c hydrogen has a carbon (C3) with four different groups: H, Cl, CH3, and CH(CH3)CH3, making it chiral.
Only one unique monochlorinated product has a chiral center. However, chiral compounds exist as enantiomers (mirror images). So, for the chiral molecule, we have a pair of enantiomers, but they are two distinct compounds.
Thus, number of chiral compounds = 2.
2 chiral compounds are possible.
Chirality arises when a molecule has a carbon atom bonded to four different groups, satisfying the condition for asymmetry. The number of stereoisomers for a molecule with n chiral centers can be up to , but in this case, n=1, so 2 enantiomers.
In monochlorination, the product distribution depends on the number of equivalent hydrogens and the reactivity (tertiary > secondary > primary).