Compare C–H bond energy.
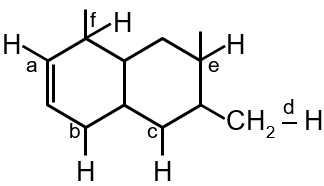
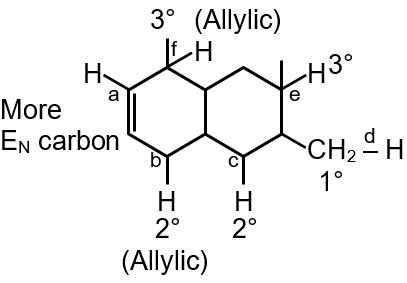
The C–H bond energy depends on the hybridization state of the carbon atom and the stability of the resulting radical after bond dissociation. Higher s-character in hybridization leads to stronger bonds due to better orbital overlap and shorter bond lengths.
Different carbon atoms have different hybridizations:
From the image (assuming standard representations):
Bond energy order: sp > sp² > sp³, with modifications for radical stability:
Alkyne (a) > Benzene (d) > Alkene (c) > Allylic (e) > Alkane (b) > Benzylic (f)
Benzylic has lowest bond energy due to maximum radical stabilization:
Correct order: a > d > c > e > b > f
Bond Dissociation Energy: Energy required to break a bond homolytically. Affected by hybridization and radical stability.
Radical Stability: Tertiary > Secondary > Primary > Methyl, with additional stabilization through resonance.
Bond energy generally correlates with s-character:
For benzylic/allylic systems: