Bromination of cyclohexene under conditions given below yields :
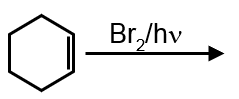
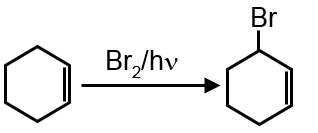
It is free radical substitution reaction.
Cyclohexene is an alkene that undergoes bromination via an electrophilic addition mechanism. The reaction involves the addition of bromine (Br2) across the carbon-carbon double bond, resulting in the formation of a dibromide product. Under standard conditions (e.g., in an inert solvent like CCl4), this reaction proceeds through a bromonium ion intermediate, leading to anti-addition.
Step 1: The π electrons of the double bond in cyclohexene attack Br2, forming a cyclic bromonium ion intermediate and a bromide ion (Br–).
Step 2: The bromide ion attacks the bromonium ion from the opposite side (anti-addition), resulting in trans-1,2-dibromocyclohexane. The substituents are added to opposite faces of the double bond.
Final Answer: The product is trans-1,2-dibromocyclohexane, where the bromine atoms are in a diaxial or diequatorial conformation, but typically represented with anti stereochemistry. Among the options, the correct structure is the one showing two bromines on adjacent carbons in a trans configuration (often drawn with one wedge and one dash bond).
General reaction for bromination of alkenes:
The mechanism involves anti addition due to the bromonium ion, leading to meso or racemic mixtures in acyclic systems, but in cyclohexene, it specifically gives the trans-diaxial product initially, which may equilibrate to the diequatorial form.