For which of the following molecule significant µ ≠ 0?
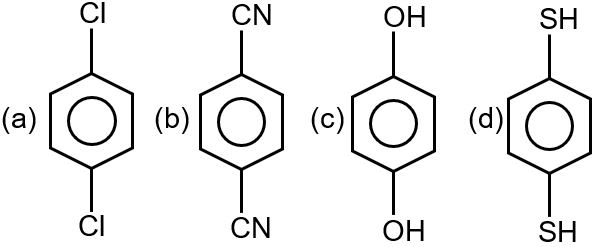
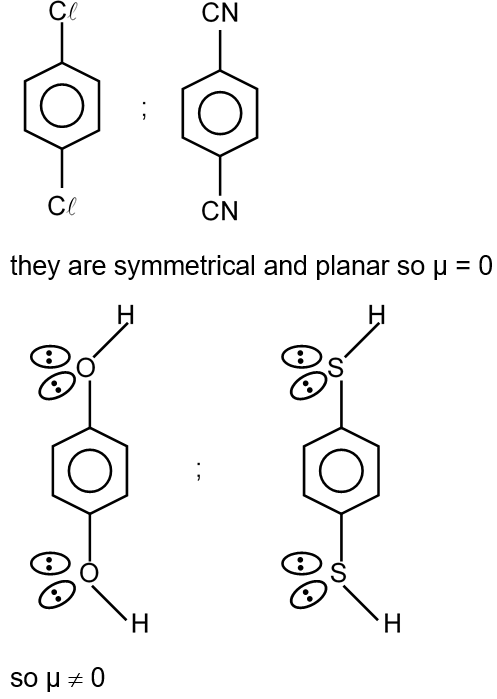
The question asks: "For which of the following molecule significant μ ≠ 0?" This means we need to identify which molecules have a permanent dipole moment. A molecule has a permanent dipole moment (μ ≠ 0) if it is polar, which occurs when there is a net separation of positive and negative charges due to differences in electronegativity and the molecule's geometry is not symmetric.
Step 1: Analyze Molecule (a)
Molecule (a) is 1,3-dichlorobenzene. It has a symmetric structure due to the para-substitution of chlorine atoms. The dipole moments of the two C-Cl bonds are equal in magnitude but opposite in direction, so they cancel each other out.
Result: Net dipole moment, μ = 0.
Step 2: Analyze Molecule (b)
Molecule (b) is 1,4-dichlorobenzene. Similar to (a), it is para-substituted and symmetric. The dipole moments of the C-Cl bonds cancel each other.
Result: Net dipole moment, μ = 0.
Step 3: Analyze Molecule (c)
Molecule (c) is 1,2-dichlorobenzene (ortho-dichlorobenzene). The chlorine atoms are adjacent, creating an asymmetric structure. The dipole moments do not cancel; they add up due to their directions.
Result: Net dipole moment, μ ≠ 0.
Step 4: Analyze Molecule (d)
Molecule (d) is 1,2,4-trichlorobenzene. The chlorine atoms are not symmetrically placed. The unequal distribution and angles prevent cancellation of dipole moments.
Result: Net dipole moment, μ ≠ 0.
Molecules (c) and (d) have significant μ ≠ 0. Therefore, the correct option is (c) and (d).
The dipole moment μ for a bond is given by:
where is the magnitude of charge separation and is the distance between charges.
For a molecule, the net dipole moment is the vector sum of all individual bond dipole moments. If the vector sum is zero, the molecule is non-polar (μ = 0); otherwise, it is polar (μ ≠ 0).
Example: In CO₂, the bond dipoles cancel due to linear symmetry, so μ = 0. In H₂O, the bent shape leads to a net dipole, so μ ≠ 0.