Understanding Permanent Dipole Moment
A molecule has a permanent dipole moment if it has polar bonds (due to electronegativity differences) and an asymmetric shape (net dipole doesn't cancel out). Symmetric molecules with polar bonds may have zero dipole moment.
Step 1: Analyze Each Option
Option 1: BeCl2, CO2, BCl3, CHCl3
- BeCl2: Linear (symmetrical),
- CO2: Linear (symmetrical),
- BCl3: Trigonal planar (symmetrical),
- CHCl3: Tetrahedral (asymmetric),
Not all have permanent dipole.
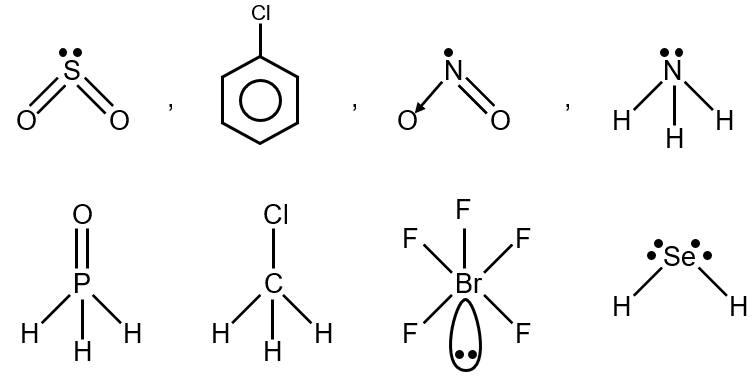
Option 2: SO2, C6H5Cl, H2Se, BrF5
- SO2: Bent shape (asymmetric),
- C6H5Cl (Chlorobenzene): Polar C-Cl bond, asymmetric,
- H2Se: Bent shape (asymmetric),
- BrF5: Square pyramidal (asymmetric),
All have permanent dipole.
Option 3: BF3, O3, SF6, XeF6
- BF3: Trigonal planar (symmetrical),
- O3: Bent shape (asymmetric),
- SF6: Octahedral (symmetrical),
- XeF6: Distorted octahedral (asymmetric),
Not all have permanent dipole.
Option 4: NO2, NH3, POCl3, CH3Cl
- NO2: Bent shape (asymmetric),
- NH3: Pyramidal (asymmetric),
- POCl3: Tetrahedral (asymmetric due to different atoms),
- CH3Cl: Tetrahedral (asymmetric),
All have permanent dipole.
Step 2: Final Answer
Options where all four molecules possess permanent dipole moment are: Option 2 and Option 4.
Related Topics & Formulae
Key Concepts:
- Polar Bonds: Occur when atoms with different electronegativities bond. Electronegativity difference usually indicates polarity.
- Molecular Geometry: Determines if bond dipoles cancel. Symmetrical shapes (linear, trigonal planar, tetrahedral with identical atoms, etc.) often have .
- Dipole Moment (): Vector sum of individual bond dipoles. , where is charge magnitude and is distance.
Common Symmetric (Zero Dipole) Molecules: BeCl2, CO2, BF3, CH4, CCl4, SF6, etc.
Common Asymmetric (Non-zero Dipole) Molecules: H2O, SO2, NH3, CHCl3, CH3Cl, O3, etc.