In the Hofmann bromamide degradation reaction, the number of moles of NaOH and Br2 used per mole of amine produced are :
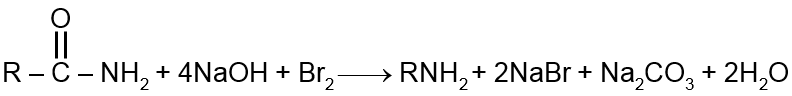
The Hofmann bromamide degradation reaction is a method to convert a primary amide into a primary amine with one less carbon atom. The reaction involves the treatment of an amide with bromine and sodium hydroxide.
The general reaction is:
Let's break down the stoichiometry:
Step 1: The amide (RCONH2) reacts with bromine (Br2) in the presence of a base (NaOH) to form an intermediate isocyanate (RNCO).
Step 2: The isocyanate is hydrolyzed by the base to yield the primary amine (RNH2).
From the balanced chemical equation, we can see that for every 1 mole of primary amine (RNH2) produced:
Therefore, the correct answer is Four moles of NaOH and one mole of Br2.
Amines: Amines are organic compounds derived from ammonia (NH3) by replacing one or more hydrogen atoms with alkyl or aryl groups. Primary amines (RNH2) have one alkyl/aryl group attached to the nitrogen atom.
Amides: Amides are carboxylic acid derivatives with the general formula RCONH2. They are less basic than amines due to the resonance effect of the carbonyl group.
The general formula for the Hofmann bromamide degradation reaction is: