Silver Mirror test is given by which one of the following compounds ?
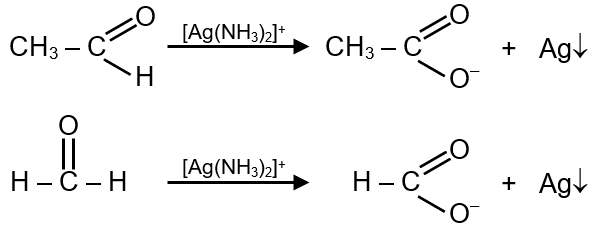
The Silver Mirror Test (also known as Tollens' test) is a chemical test used to distinguish between aldehydes and ketones. It involves the oxidation of aldehydes to carboxylic acids, while reducing silver ions (Ag⁺) to metallic silver (Ag), which forms a shiny mirror-like coating on the test tube.
Step 1: Understand the reaction - Tollens' reagent (ammoniacal silver nitrate, [Ag(NH₃)₂]⁺) oxidizes aldehydes but not ketones.
Step 2: The general reaction for an aldehyde (RCHO) is:
Step 3: Analyze the options:
Final Answer: Acetaldehyde gives the silver mirror test.
Tollens' Reagent Preparation: AgNO₃ + NaOH → Ag₂O (precipitate) + NH₄OH → [Ag(NH₃)₂]⁺
Key Theory: Aldehydes are easily oxidized to carboxylic acids because they have a hydrogen atom attached to the carbonyl carbon, while ketones do not have this hydrogen and are resistant to mild oxidation.
General Reaction: R-CHO + 2[Ag(NH₃)₂]⁺ + 3OH⁻ → R-COO⁻ + 2Ag + 4NH₃ + 2H₂O