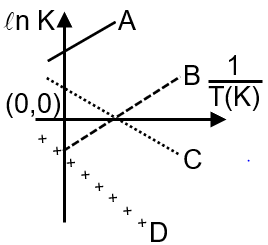
Which of the following lines correctly show the temperature dependence of equilibrium constant, K, for an exothermic reaction?
ln (K) =
For exothermic reaction H° < 0
so slope will be +ve
A and B are correct
The equilibrium constant (K) for a reaction is related to temperature by the van't Hoff equation:
For an exothermic reaction, the standard enthalpy change is negative. This means:
This inequality tells us that ln K decreases as temperature T increases. Since ln K is a monotonically increasing function of K, this also means the equilibrium constant K itself decreases as temperature increases.
Now let's analyze the graph options. The x-axis is temperature (T) and the y-axis is the equilibrium constant (K).
Step 1: Identify the decreasing curves
Since K must decrease with increasing T for an exothermic reaction, we are looking for curves that slope downward from left to right. In the image, curves A and D are decreasing.
Step 2: Verify the mathematical behavior
The van't Hoff equation also provides the integrated form for a constant : This equation has the form y = mx + c, where y = ln K and x = 1/T. For an exothermic reaction (negative slope m), a plot of ln K vs 1/T is a straight line with a positive slope (because of the negative sign in front of the negative ). However, the given graph is K vs T, not ln K vs 1/T. The relationship between K and T is not linear but follows the form above, resulting in a curve that decreases, like A and D.
Final Answer: The lines that correctly show the temperature dependence for an exothermic reaction are A and D.
van't Hoff Equation: This equation is fundamental for understanding how equilibrium constants change with temperature. It is derived from the relationship between the Gibbs free energy change and the equilibrium constant: and the Gibbs-Helmholtz equation: .
Le Chatelier's Principle: This qualitative principle predicts that for an exothermic reaction (which releases heat), increasing the temperature will shift the equilibrium position to the left, towards the reactants. This shift results in a decrease in the equilibrium constant K, which is a quantitative measure of the position of equilibrium. This principle is consistent with the mathematical result from the van't Hoff equation.