Which of the following facts about the complex [Cr(NH3)6] Cl3 is wrong ?
In case of d3 configuration, the number of unpaired electrons remains 3 whether the ligand is strong field or weak field. The hybridisation scheme can be shown as follow :
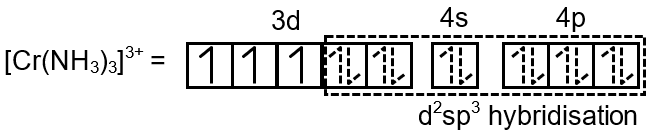
Hence the complex is inner orbital complex as it involves (n – 1) d orbitals for hybridisation, ; so n = 3 (here n is number of unpaired electron(s)).
The complex given is [Cr(NH3)6]Cl3. To determine which fact is wrong, we need to analyze its hybridization, magnetic behavior, and precipitation reaction.
Step 1: Determine the oxidation state of chromium.
Let the oxidation state of Cr be x. Since NH3 is neutral, and the overall charge is +3 (because of three Cl- ions), we have:
So, x = +3. Thus, Cr is in +3 oxidation state.
Step 2: Write the electronic configuration of Cr3+.
Atomic number of Cr is 24. Electronic configuration of Cr is [Ar] 4s1 3d5.
For Cr3+, it loses 3 electrons. So, configuration is [Ar] 3d3.
Step 3: Determine hybridization and orbital type.
NH3 is a strong field ligand. For Cr3+ (d3), in an octahedral field, the electrons pair up? Actually, d3 has 3 unpaired electrons. But with strong field ligands, it can undergo inner orbital hybridization (d2sp3). Since there are 3 electrons in d orbitals, they occupy three orbitals, leaving two d orbitals empty. So, hybridization is d2sp3, which is inner orbital. Thus, it is not an outer orbital complex (outer orbital would be sp3d2).
Step 4: Check magnetic behavior.
With 3 unpaired electrons, the complex is paramagnetic.
Step 5: Check precipitation with AgNO3.
The complex is [Cr(NH3)6]Cl3, so it dissociates to give three Cl- ions. These will form white precipitate with AgNO3.
Conclusion:
- It involves d2sp3 hybridization and is octahedral: Correct.
- It gives white precipitate with AgNO3: Correct.
- It is paramagnetic: Correct.
- It is an outer orbital complex: Wrong, because it is inner orbital (d2sp3).
Therefore, the wrong fact is that it is an outer orbital complex.
Hybridization in Coordination Compounds:
For octahedral complexes, hybridization can be d2sp3 (inner orbital) or sp3d2 (outer orbital). Inner orbital is preferred when the metal ion has empty inner d orbitals and ligands are strong field.
Magnetic Behavior:
Paramagnetic if unpaired electrons are present; diamagnetic if all paired.
Precipitation Reaction:
Cl- ions form AgCl (white precipitate) with Ag+.