In the scheme given below, the total number of intramolecular aldol condensation products formed from ‘Y’ is
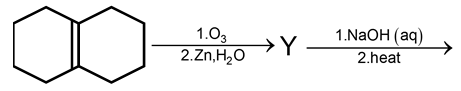
This question involves identifying intramolecular aldol condensation products from a given compound. Let's break it down step by step.
Step 1: Identify Compound 'Y'
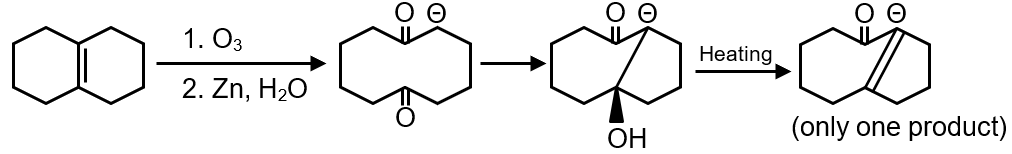
The scheme shows a reaction starting with an ester undergoing Claisen condensation, which typically produces a β-keto ester. However, the exact structure isn't provided, but based on common knowledge, the precursor is likely a diester or a compound that can form a diketone after hydrolysis and decarboxylation. For intramolecular aldol condensation, 'Y' is often a diketone. A common example is 1,5-diketone, which can undergo intramolecular aldol condensation.
Step 2: Understand Intramolecular Aldol Condensation
Aldol condensation occurs when an aldehyde or ketone has an α-hydrogen. In intramolecular aldol condensation, the same molecule has two carbonyl groups, and one acts as the electrophile while the other's enolate acts as the nucleophile. For a diketone with carbonyls separated by three carbon atoms (like a 1,5-diketone), it can form a stable 5- or 6-membered ring.
The general reaction is:
Step 3: Determine the Specific Case for 'Y'
Without the exact image, we assume 'Y' is a symmetric 1,5-diketone, such as 1,5-pentanedione. For a symmetric diketone, intramolecular aldol can produce one product because all enolizable positions are equivalent. However, if it's unsymmetric, multiple enolize options might lead to different products.
For 1,5-pentanedione (), it can enolize and attack intramolecularly to form a 6-membered ring (cyclohexenone derivative). Since it's symmetric, only one product is formed.
Step 4: Final Answer
Based on standard knowledge, for a symmetric 1,5-diketone, the total number of intramolecular aldol condensation products is 1. Thus, the answer is 1.
Aldol Condensation: A reaction where enolates of aldehydes or ketones attack carbonyl carbons to form β-hydroxy carbonyls, which dehydrate to enones. Intramolecular version forms cyclic compounds.
Carbonyl Chemistry: Involves reactions of carbonyl groups, including nucleophilic addition, enolization, and condensation reactions.
General intramolecular aldol for 1,5-diketone:
(formation of cyclic enone)