Which is the major product formed when acetone is heated with iodine and potassium hydroxide?
This question involves the iodoform test, a specific chemical reaction used to identify methyl ketones (like acetone) and compounds that can be oxidized to methyl ketones (like ethanol).
Step 1: Understanding the Reaction
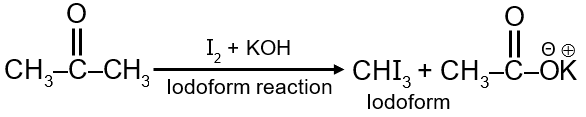
When a methyl ketone (a ketone with the structure R-CO-CH3) is treated with iodine (I2) and a base (like potassium hydroxide, KOH), it undergoes a haloform reaction. The base first facilitates the alpha-halogenation of the ketone. Since acetone has three equivalent alpha-hydrogens, all three can be successively replaced by iodine atoms.
Step 2: The Mechanism
The reaction proceeds through the following key steps:
Final Answer: The major product is a yellow precipitate of iodoform (CHI3). The other product is acetate ion, which would form acetic acid in an acidic workup.
Why the other options are incorrect:
Haloform Reaction General Equation:
For a methyl ketone: R-CO-CH3 + 3X2 + 4NaOH → R-COO-Na+ + CHX3 + 3NaX + 3H2O
(Where X can be Cl, Br, or I)
Compounds that give a positive iodoform test: Acetaldehyde, Ethanol, and any methyl ketone.