Under hydrolytic conditions, the compounds used for preparation of linear polymer and for chain termination, respectively, are
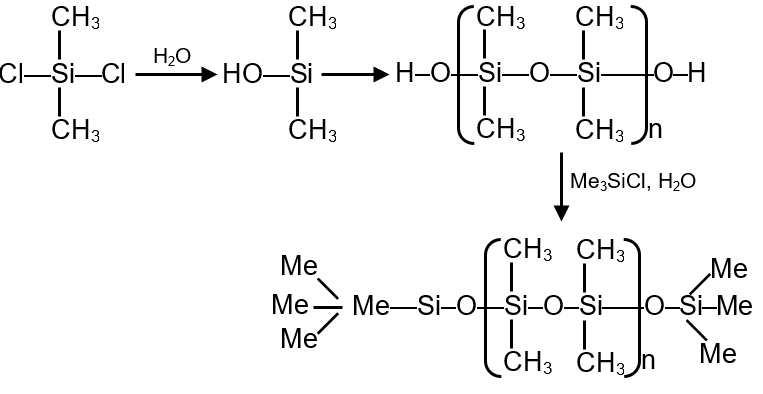
Silicones are synthetic polymers with a backbone of alternating silicon and oxygen atoms, with organic groups attached to silicon. They are prepared by hydrolysis of chlorosilanes. The type of chlorosilane used determines the polymer structure:
Step 1: Identify the compound that forms linear polymer chains. This requires a bifunctional monomer: (CH3)2SiCl2.
Hydrolysis:
Step 2: Identify the chain termination agent. This requires a monofunctional compound: (CH3)3SiCl.
Termination reaction:
Step 3: Evaluate the options:
Final Answer: The correct pair is (CH3)2SiCl2 and (CH3)3SiCl.
Key Concepts:
Applications: Silicones are used in sealants, lubricants, medical devices, and cookware due to their thermal stability, water repellency, and flexibility.