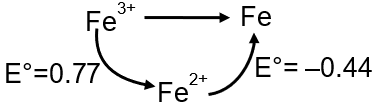
The standard reduction potential data at 25°C is given below.
E° (Fe3+, Fe2+) = +0.77 V ;
E° (Fe2+, Fe) = –0.44 V
E° (Cu2+, Cu) = +0.34 V;
E° (Cu+, Cu) = + 0.52 V
E° [O2(g) + 4H+ + 4e– → 2H2O] = +1.23 V;
E° [O2(g) + 2H2O + 4e– → 4OH¯] = +0.40 V
E° (Cr3+, Cr) = – 0.74 V;
E°(Cr2+, Cr) = –0.91 V
Match E° of the redox pair in List I with the values given in List II and select the correct answer using the code given below the lists:
| List I | List II |
| (P) E° (Fe3+, Fe) | (1) – 0.18 V |
| (Q) E° (4H2O ⇌ 4H+ + 4OH¯) | (2) – 0.4 V |
| (R) E° (Cu2+ + Cu → 2Cu+) | (3) – 0.04 V |
| (S) E° (Cr3+, Cr2+) | (4) – 0.83 V |
Checking easiest option P or S
Cr3+ → Cr2+
E° = – 0.74 × 3 + 0.91 × 2
= – 0.4 V
We are given standard reduction potentials for several half-cells and need to find the standard reduction potentials for four specific redox pairs: (P) Fe3+/Fe, (Q) 4H2O ⇌ 4H+ + 4OH–, (R) Cu2+ + Cu → 2Cu+, and (S) Cr3+/Cr2+. We must match them to their correct values from List II.
The key concept is that the standard reduction potential for a reaction that is a combination of other half-reactions can be found by relating the Gibbs free energy changes, since . The free energy is an additive property.
The reduction can be obtained by adding two known half-reactions:
1. ;
2. ;
Adding these gives the desired reaction: ;
Therefore,
Solving for EP°:
Match: (P) with (3) –0.04 V
This represents the autoionization of water. The reaction is the reverse of the reduction reaction given for acidic and basic media:
Given: ; E° = +1.23 V
Given: ; E° = +0.40 V
The difference between these two reactions is precisely the autoionization reaction (Q) multiplied by 4. Subtracting the second reaction from the first:
Simplifying: (This is the reverse of reaction Q)
The E° for the reaction as written in List I (4H2O ⇌ 4H+ + 4OH–) is found from the relationship between the Gibbs free energies:
Therefore,
Match: (Q) with (4) –0.83 V