The packing efficiency of the two‑dimensional square unit cell shown below is
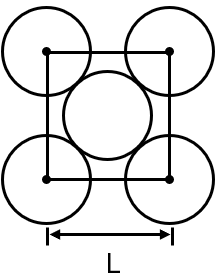
Area of square = L2
% packing efficiency
The packing efficiency of a two-dimensional square unit cell refers to the percentage of the total area that is occupied by the atoms (or particles) within the unit cell. For the given square unit cell, we need to identify the number of atoms per unit cell and their effective area to calculate the packing efficiency.
Step 1: Analyze the unit cell structure.
The image shows a square unit cell with atoms at each corner. In a 2D square lattice, each corner atom is shared by four adjacent unit cells. Therefore, the contribution of each corner atom to one unit cell is 1/4.
Step 2: Calculate the number of atoms per unit cell.
Number of corner atoms = 4
Effective number of atoms per unit cell, atom.
Step 3: Relate the atom radius to the unit cell edge length.
In a square unit cell with atoms touching along the edge, the edge length is equal to twice the radius of the atom. So, .
Step 4: Calculate the total area occupied by atoms.
Area of one atom =
Total occupied area =
Step 5: Calculate the total area of the unit cell.
Area of unit cell =
Step 6: Calculate packing efficiency.
Packing efficiency =
Since ,
Packing efficiency =
Therefore, the packing efficiency is 78.54%, which matches the third option.
Packing efficiency is a key concept in solid state chemistry, particularly in understanding the arrangement of atoms in crystals. It is directly related to the density and stability of the crystal structure. For three-dimensional structures, such as simple cubic, body-centered cubic, and face-centered cubic, the packing efficiencies differ and are calculated similarly by considering the volume instead of area.
For a 2D square unit cell:
Number of atoms per unit cell,
Edge length,
Packing efficiency =
For a 2D hexagonal close packing (which has higher efficiency), the packing efficiency is about 90.69%.