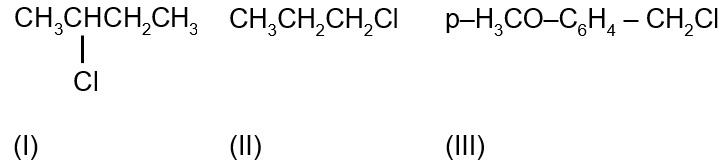
The increasing order of the reactivity of the following halides for the SN1 reaction is:
The SN1 reaction (unimolecular nucleophilic substitution) proceeds through a carbocation intermediate. The rate of SN1 reaction depends on the stability of the carbocation formed. More stable the carbocation, faster is the SN1 reaction.
Let's analyze the carbocations formed from each halide:
Compound (I): Benzyl chloride. Upon ionization, it forms benzyl carbocation . This carbocation is stabilized by resonance with the benzene ring. The positive charge is delocalized into the ring, making it very stable.
Compound (II): p-Chloro benzyl chloride. The -Cl group is electron-withdrawing due to its -I effect. This group destabilizes the positive charge on the benzylic carbon. The resonance structures that delocalize the charge into the ring are less effective because the ring itself is made electron-deficient by the chlorine substituent. Therefore, the carbocation from (II) is less stable than the carbocation from (I).
Compound (III): p-Methoxy benzyl chloride. The -OCH3 group is electron-donating due to its +M and +I effects. This group stabilizes the positive charge on the benzylic carbon. The resonance structures that delocalize the charge into the ring are enhanced because the ring is made electron-rich by the methoxy substituent. In fact, the methoxy group can donate electrons directly into the carbocation through resonance. Therefore, the carbocation from (III) is the most stable among the three.
Order of Carbocation Stability: (III) > (I) > (II)
Since SN1 rate ∝ Carbocation stability, the order of reactivity for SN1 reaction is: (III) > (I) > (II)
Therefore, the increasing order of reactivity is: (II) < (I) < (III)
SN1 Reaction Mechanism: A two-step process where the rate-determining step is the unimolecular dissociation of the alkyl halide to form a carbocation, followed by a fast attack by the nucleophile.
Carbocation Stability Order: 3° > 2° > 1° > methyl. Furthermore, resonance stabilization (e.g., allylic, benzylic) greatly increases stability.
Effects of Substituents on Benzene Ring: Electron-donating groups (e.g., -OCH3, -OH) stabilize positive charge (ortho/para directors). Electron-withdrawing groups (e.g., -Cl, -NO2) destabilize positive charge (meta directors).