The colour of light absorbed by an aqueous solution of CuSO4 is
aqueos solution of CuSO4 is blue so absorbed colour is orange-red
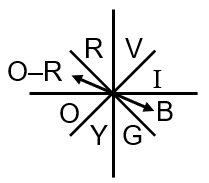
Copper sulfate (CuSO₄) solution appears blue because of its light absorption properties. The color we see is the complementary color to the one absorbed.
The blue color comes from hydrated copper ions in solution.
These ions absorb light in the orange-red region of the visible spectrum (approximately 600-800 nm wavelength).
Since orange-red light is absorbed, the transmitted light appears as its complementary color. The complementary color to orange-red is blue-green, but in practice, the solution appears blue due to the specific wavelengths involved.
The color of light absorbed by an aqueous solution of CuSO₄ is Orange-red.
Beer-Lambert Law:
Where A is absorbance, ε is molar absorptivity, c is concentration, and l is path length.
Complementary Colors: Colors opposite each other on the color wheel are complementary. When a substance absorbs one color, we perceive the complementary color.
Crystal Field Theory: Explains how d-d transitions in transition metal complexes lead to color absorption through splitting of d-orbitals.