Sodium salt of an organic acid 'X' produces effervescence with conc. H2SO4, 'X' reacts with the acidified aqueous CaCl2 solution to give a white precipitate which decolourises acidic solution of KMnO4. 'X' is:
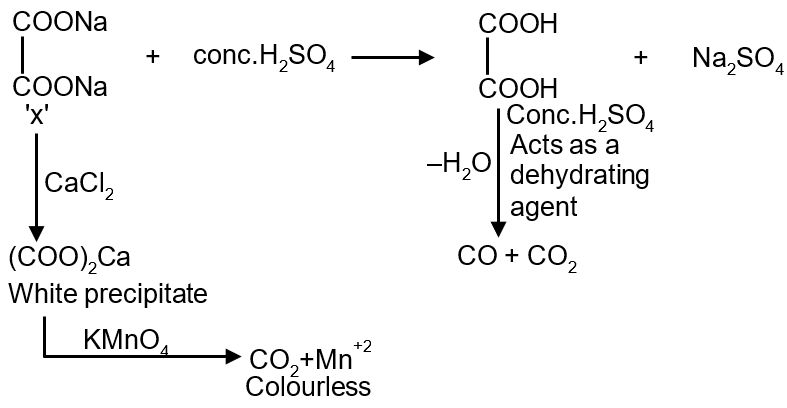
This question involves identifying an organic acid 'X' based on its sodium salt's reactions. Let's analyze the clues step by step:
Sodium salts of organic acids produce CO2 gas (effervescence) when treated with concentrated H2SO4 due to decomposition. This is a general test for carboxylate salts, but it doesn't distinguish between them.
'X' forms a white precipitate with acidified CaCl2 solution. This indicates the formation of an insoluble calcium salt. Common organic acids that form insoluble calcium salts include oxalic acid (calcium oxalate) and formic acid (calcium formate is somewhat soluble, but can precipitate under certain conditions). However, calcium oxalate (CaC2O4) is highly insoluble and a characteristic precipitate.
The white precipitate decolorizes acidic potassium permanganate (KMnO4) solution. This is a crucial test. Acidic KMnO4 is decolorized by reducing agents. Oxalate ion (C2O42-) is a strong reducing agent and reduces MnO4- to Mn2+, decolorizing the purple solution. The reaction is:
Formate ion (HCOO-) also reduces acidic KMnO4, but it does not form a highly characteristic white precipitate with CaCl2 (calcium formate is moderately soluble). Acetate and benzoate ions do not reduce KMnO4 and their calcium salts are soluble.
Combining all clues:
Tests for Carboxylic Acids and Their Salts:
Oxidizing Agents:
Important Reaction:
The oxidation of oxalate ion by permanganate is a classic volumetric reaction used to standardize KMnO4 solutions.