Select the species having marked Bond angle > 109°.
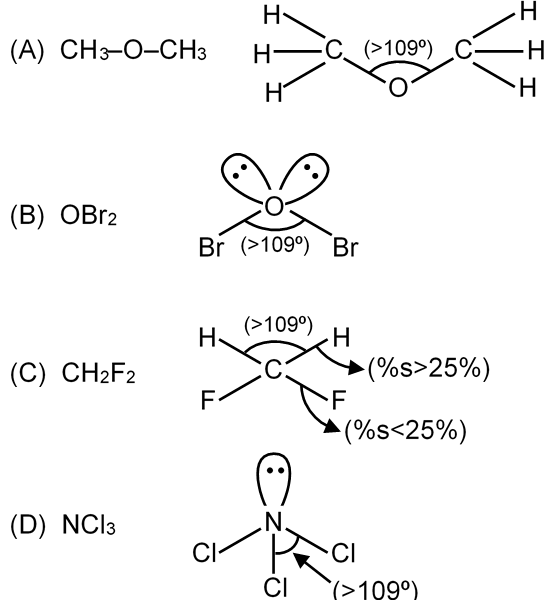
Bond angles are influenced by electron pair repulsions in molecules. The tetrahedral angle is 109.5°, but deviations occur due to lone pairs or electronegativity differences. Angles >109° typically occur when:
Option 1: Dimethyl ether (CH3-O-CH3)
Central atom: Oxygen (O) with 2 bond pairs and 2 lone pairs. Lone pair-bond pair repulsion reduces the bond angle from tetrahedral. Expected angle ≈ 104.5° (less than 109°).
Option 2: OBr2
Central atom: Oxygen (O) with 2 bond pairs and 2 lone pairs. Similar to water (H2O), bond angle ≈ 103° (less than 109°).
Option 3: NCl3
Central atom: Nitrogen (N) with 3 bond pairs and 1 lone pair. Lone pair repulsion reduces angle from tetrahedral to ≈ 107° (less than 109°).
Option 4: CH2F2
Central atom: Carbon (C) with 4 bond pairs (no lone pairs). Tetrahedral geometry, but fluorine is highly electronegative, pulling electron density and increasing H-C-H repulsion. Bond angle >109.5° (typically ≈ 112°).
Only CH2F2 has no lone pairs and high electronegativity difference, leading to angle >109°.
CH2F2 has a bond angle >109°.
Bond angle (θ) in AXnEm systems:
where m is number of lone pairs, and δ is positive for electronegative substituents.