Which properties is related to given graph?
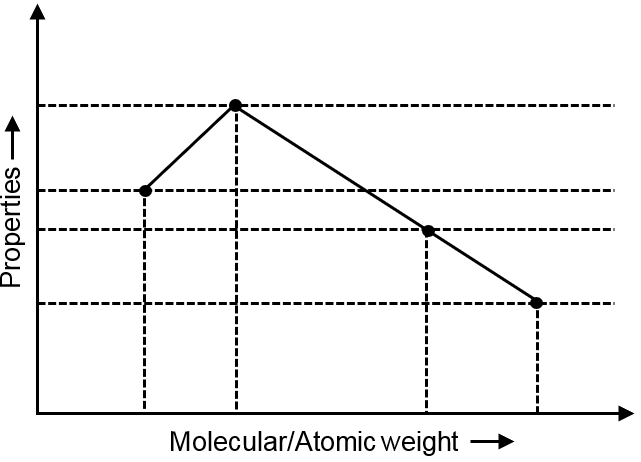
(A) Electron affinity of group-16 elements.→ S > Se > Te > O
(B) Bond energy of halogen. → Cl2 > Br2 > F2 > I2
(C) Lewis basic nature of group 15 hydride. → NH3 > PH3 > AsH3 > SbH3
(D) Electron affinity of group 17 elements. → Cl > F > Br > I
The graph shows a decreasing trend followed by an increase, forming a "V" shape pattern. This pattern is characteristic of properties that decrease down a group but show an anomaly at a particular element.
The graph shows values decreasing from first to second element, then increasing for subsequent elements. This indicates an exception to the general periodic trend.
Among the options:
The V-shaped pattern matches perfectly with the Lewis basic nature of group 15 hydrides:
NH₃ > PH₃ < AsH₃ < SbH₃ < BiH₃
This anomaly occurs due to the small size and high electronegativity of nitrogen in NH₃.
Lewis basic nature of group 15 hydride is the property related to the given graph.
Lewis basicity is the ability to donate an electron pair. For group 15 hydrides:
Basicity ∝
However, NH₃ shows exceptional basicity due to:
The basic strength order is: NH₃ > PH₃ < AsH₃ < SbH₃ < BiH₃