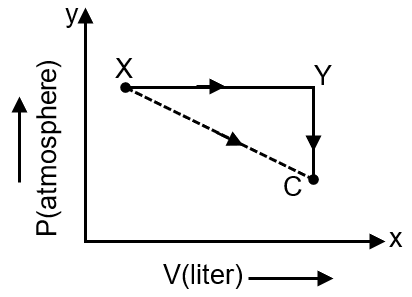
For an ideal gas, consider only P-V work in going from an initial state X to the final state Z. The final state Z can be reached by either of the two paths shown in the figure. Which of the following choice(s) is (are) correct ? [take ΔS as change in entropy and was work done]
No Solution
We are analyzing an ideal gas undergoing a process from state X to state Z via two different paths: one direct path (X→Z) and one indirect path (X→Y→Z). The work done (W) and change in entropy (ΔS) are path-dependent and state functions respectively. We need to determine which of the given options are correct.
Work done in a P-V process is given by . Work is a path function, meaning it depends on the path taken.
For path X→Y→Z, the total work done is the sum of work in each step: . However, the direct path X→Z may have a different work value. Since the paths are different, in general. Looking at the figure, path X→Y is at constant volume (isochoric), so . Path Y→Z is at constant pressure (isobaric), so . Therefore, . The direct path X→Z is a straight line, and its work is the area under the curve, which is different from the area under the constant pressure path Y→Z. Hence, and also is true by definition, but is not necessarily true.
Entropy is a state function. The change in entropy depends only on the initial and final states, not on the path taken. Therefore, for any path from X to Z, is the same.
For the indirect path X→Y→Z, the total entropy change is . Since entropy is a state function, this sum must equal the entropy change for the direct path: . Therefore, is correct.
However, is incorrect because it ignores the entropy change from Y to Z.
Option 1:
This is false. Work is path-dependent, and the work for the direct path X→Z is not equal to the sum of works for path X→Y and Y→Z.
Option 2:
This is false. The total entropy change for path X→Y→Z includes both steps, not just the first.
Option 3:
This is true. Entropy is a state function, so the change is additive over paths.
Option 4:
This is false. The total work for path X→Y→Z is not just the work from X→Y (which is zero) but also includes the work from Y→Z.
Only Option 3 is correct:
State Functions: Properties that depend only on the initial and final states of the system (e.g., Internal Energy (U), Enthalpy (H), Entropy (S), Gibbs Free Energy (G)).
Path Functions: Properties that depend on the path taken between initial and final states (e.g., Work (W), Heat (Q)).
For a reversible process, work done by the gas is given by: