An ideal gas in a thermally insulated vessel at internal pressure = P1, volume = V1 and absolute temperature = T1 expands irreversibly against zero external pressure, as shown in the diagram. The final internal pressure, volume and absolute temperature of the gas are P2 , V2 and T2 , respectively. For this expansion,
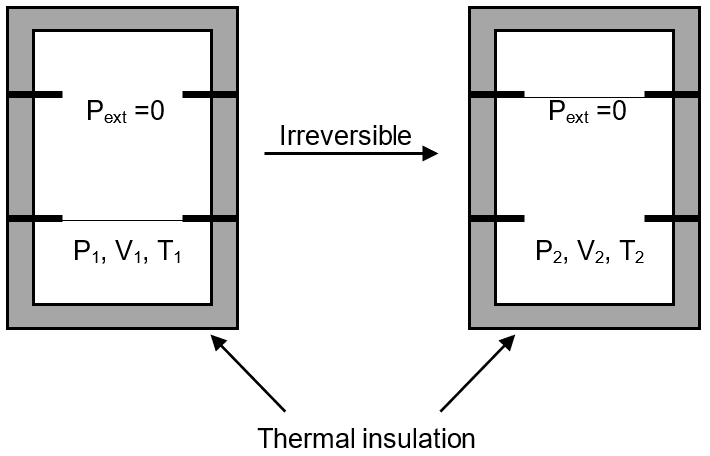
Pext = 0
w = 0 thermally insulated q = 0
∴ ΔU = 0 , hence T2 = T1
P1V1 = P2V2
This problem involves an ideal gas expanding irreversibly against zero external pressure in a thermally insulated vessel. Let's analyze the situation step by step.
The vessel is thermally insulated, meaning no heat exchange occurs with the surroundings. Therefore, . This is an adiabatic process.
The gas expands against zero external pressure (). The work done by the gas in an irreversible expansion is given by:
Since , the work done is:
The first law states:
We have already established that and . Therefore:
For an ideal gas, the internal energy depends only on temperature. If , then the temperature must remain constant.
Since the temperature is constant (), we can apply Boyle's Law for the initial and final states.
Initial state:
Final state:
Because , the right-hand sides of both equations are equal. Therefore:
The option is the condition for a reversible adiabatic process. Our process is irreversible and adiabatic, but it is also isothermal (). The relation is not valid here because it applies only to adiabatic processes where temperature changes.
For this expansion, the following statements are correct:
The statement is incorrect for this process.
An adiabatic process is one where no heat is exchanged ().
This is a unique case of an adiabatic process that is also isothermal. It only happens when an ideal gas expands against zero pressure, resulting in zero work. Since no work is done and no heat is exchanged, there is no change in internal energy, and hence, no change in temperature.