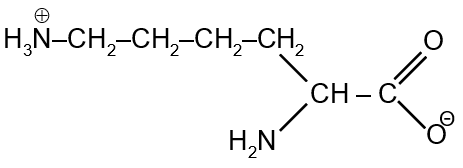
The total number of basic groups in the following form of lysine is
Lysine is an amino acid with the chemical formula C6H14N2O2. It contains an amino group (-NH2) and a carboxylic acid group (-COOH) attached to the alpha carbon, along with an additional amino group on its side chain. A basic group is one that can accept a proton (H+). In organic chemistry, amino groups (-NH2) are basic because the nitrogen atom has a lone pair that can accept a proton.
The given structure of lysine (in the image) is:
H2N-CH2-CH2-CH2-CH2-CH(NH2)-COOH
This shows lysine in its neutral form, with two amino groups: one at the alpha position (attached to the carbon next to COOH) and one at the end of the side chain.
Basic groups in amino acids are typically amino groups (-NH2). In lysine:
Note: The carboxylic acid group (-COOH) is acidic, not basic.
Lysine has two amino groups, both of which are basic. Therefore, the total number of basic groups is 2.
The total number of basic groups in the given form of lysine is .
Basic Group Definition: A group that can accept a proton (Bronsted-Lowry base). For amino acids, amino groups (-NH2) are basic:
pKa and Protonation: The pKa indicates the pH where half the group is protonated. Amino groups typically have pKa values between 9 and 10.
Lysine Structure: Chemical formula: . It is classified as a basic amino acid due to its extra amino group.