The difference in the oxidation numbers of the two types of sulphur atoms in Na2S4O6 is
O.N. S* = 0
O.N. S# = + 5
Difference = 5
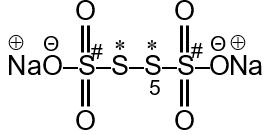
Sodium tetrathionate (Na2S4O6) contains four sulphur atoms, but they are not all equivalent. Two are central atoms with a different oxidation state than the two terminal ones. Let's find the oxidation numbers step by step.
The known oxidation numbers are:
The structure of the tetrathionate ion (S4O62-) is often represented as (O3S-S-S-SO3)2-. The two central sulphur atoms (S-S) are bonded to each other, while the two terminal sulphur atoms are each bonded to three oxygen atoms.
Let the oxidation number of each terminal S atom (the ones in -SO3 groups) be .
Let the oxidation number of each central S atom (the ones in the S-S chain) be .
The tetrathionate ion has a charge of -2. The sum of the oxidation numbers of all atoms must equal this charge.
Atoms in S4O62-: 4 S atoms and 6 O atoms.
Therefore:
This simplifies to:
Which further simplifies to:
Dividing the entire equation by 2 gives us our first key relationship:
We can find another relationship by looking at one terminal -SO3 group. This group has an overall charge that we can determine from its bonding.
In the structure, the terminal S atom is bonded to three O atoms (each with an O.N. of -2) and one central S atom. The S-S bond is a non-polar covalent bond between two identical atoms. In a non-polar covalent bond between two identical atoms, the oxidation number for each atom in that bond is zero.
Therefore, for the oxidation number calculation of the terminal S atom, the bond to the other S atom contributes 0.
The sum of oxidation numbers for the atoms in one -SO3 unit must be the charge on that group. Since it's part of an ion, we consider its oxidation number contribution. The oxidation number of the terminal S atom () plus its three O atoms () is often considered to be -2, as it is analogous to a sulphate (SO42-) group.
This gives us our second equation:
Solving for :
The oxidation number of a terminal S atom is +5.
Now, substitute the value of from Equation 2 into Equation 1.
The oxidation number of a central S atom is 0.
The question asks for the difference in the oxidation numbers of the two types of sulphur atoms.
Oxidation number of terminal S () = +5
Oxidation number of central S () = 0
The difference is:
The difference in the oxidation numbers of the two types of sulphur atoms is .
The fundamental rule for any molecule or ion:
This is the equation you set up and solve to find unknown oxidation numbers.
This concept applies to other polythionates as well (e.g., dithionate S2O62-, trithionate S3O62-). They all contain S atoms in different oxidation states, and the S-S bonds between central atoms are key to understanding their chemistry.