One mole of an ideal gas is taken from a to b along two paths denoted by the solid and the dashed lines as shown in the graph below. If the work done along the solid line path is ws and that along the dotted line path is wd, then the integer closet to the ratio wd / ws is
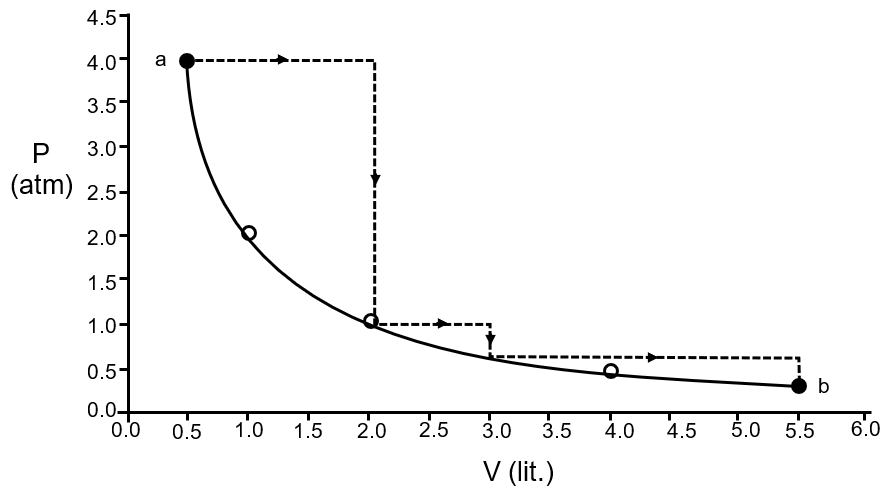
For solid line path show approxy isothermal process
∴ work done |WS| = 2.303 (PV) log
=2.303 × 4 × .5 × log 11
4.79
for dashed line path work done
wd = 4 × |2 – .5| + 1 × |3 – 2| + .5 × |5.5 –3|
= 6 + 1 + 1.25 = 8.25
This problem involves calculating work done by an ideal gas during expansion along two different paths on a P-V diagram and finding their ratio. Work done by a gas is given by the area under the P-V curve.
For any thermodynamic process, work done by the gas (W) is given by:
On a P-V diagram, this represents the area under the curve from initial to final volume.
The graph shows two paths from point a (2 L, 4 atm) to point b (6 L, 2 atm):
The dashed path consists of:
Total work for dashed path:
The solid path is a smooth curve from a to b. For an ideal gas, if this is an isothermal process (constant temperature), work done is given by:
Since n=1 mole, and using PV=nRT, we can find RT. At point a: P=4 atm, V=2 L, so:
Thus,
Since ln(3) ≈ 1.0986,
The integer closest to 1.820 is 2.
The integer closest to the ratio wd / ws is 2.
Work Done in Thermodynamic Processes:
Ideal Gas Law: PV = nRT
Graphical Interpretation: Work done is the area under the P-V curve, which depends on the path taken.