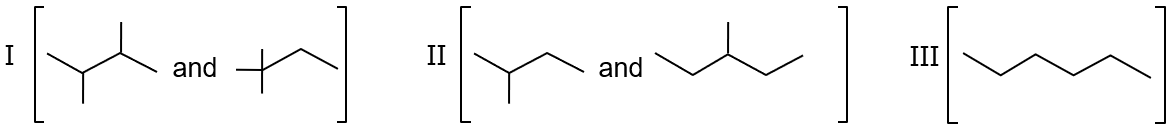
Isomers of hexane, based on their brancing, can be divided into three distinct classes as shown in the figure.
The correct order of their boiling point is
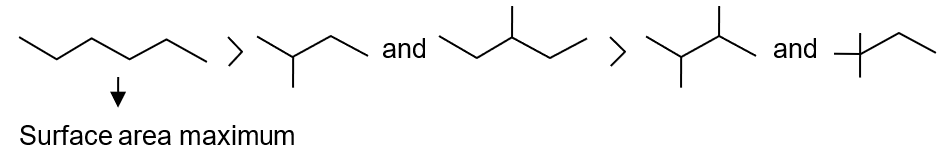
B.P. α Surface area
III > II > I
Isomers are compounds with the same molecular formula but different structural arrangements. For hexane (C6H14), branching affects physical properties like boiling point. The boiling point depends on the surface area available for intermolecular forces (van der Waals forces). More branching reduces the surface area, weakening these forces and lowering the boiling point.
Let's analyze the three isomers:
Isomer I is the straight-chain hexane (no branching). It has the largest surface area and the strongest intermolecular forces, so it has the highest boiling point.
Isomer II has one methyl branch (2-methylpentane or 3-methylpentane). This branching reduces the surface area compared to Isomer I, so its boiling point is lower than I but higher than a more branched isomer.
Isomer III is highly branched (2,2-dimethylbutane). It has the smallest surface area and the weakest intermolecular forces, so it has the lowest boiling point.
Therefore, the order of boiling points is: I > II > III (straight-chain > mono-branched > highly branched).
Structural Isomerism: Compounds with the same molecular formula but different connectivity of atoms. Hexane isomers are examples of structural isomers.
Intermolecular Forces: Forces between molecules, such as van der Waals forces, which influence physical properties like boiling point. Larger surface area leads to stronger forces.
No specific formula, but the general principle is: Boiling point ∝ Surface area (for similar molecular weight compounds). More branching ⇒ lower surface area ⇒ lower boiling point.