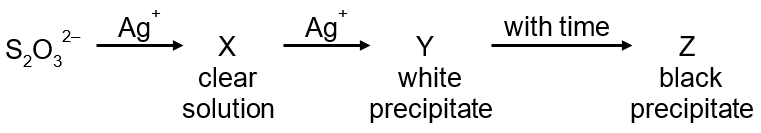
In the following reaction sequence in aqueous solution, the species X, Y and Z, respectively, are
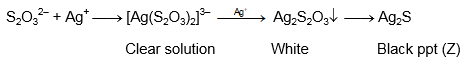
Given the reaction sequence in aqueous solution:
Ag⁺ + S₂O₃²⁻ → X (colorless)
X + S₂O₃²⁻ → Y (colorless)
Y + H₂O → Z (black)
Let's analyze each step to identify the species X, Y, and Z.
Silver ions (Ag⁺) react with thiosulfate ions (S₂O₃²⁻). Thiosulfate is a well-known complexing agent for silver. The initial reaction forms a soluble, colorless complex.
The stoichiometry of the stable silver-thiosulfate complex is well-established. The reaction is:
Therefore, the species X is the colorless complex ion [Ag(S₂O₃)₂]³⁻.
The complex X can further react with an additional thiosulfate ion. However, the complex [Ag(S₂O₃)₂]³⁻ is already quite stable. Adding more thiosulfate does not change the coordination number or form a new compound; it simply establishes an equilibrium with a very small amount of a higher complex like [Ag(S₂O₃)₃]⁵⁻, which is also colorless.
In many textbook sequences, this step is often interpreted as the formation of the same or a very similar complex for the purpose of the subsequent hydrolysis reaction. The key point is that Y is a silver-thiosulfate complex. Given the options, the most consistent and stable complex formed with excess thiosulfate is [Ag(S₂O₃)₂]³⁻ (i.e., Y is the same as X) or its equivalent. The product Y remains colorless.
Upon closer analysis of the final step (hydrolysis to a black precipitate), the initial complex X is sufficient. The addition of more thiosulfate might simply be to ensure complete complexation before the hydrolysis test. For the purpose of identifying Z, Y is effectively the silver-thiosulfate complex.
The silver-thiosulfate complex is not stable indefinitely in aqueous solution. It undergoes slow hydrolysis (reaction with water).
The complex decomposes to produce silver sulfide (Ag₂S), which is a characteristic black precipitate. This reaction is a standard test to distinguish thiosulfate from other ions like sulfate.
The hydrolysis reaction can be simplified as:
Therefore, the black species Z is Ag₂S (silver sulfide).
Based on the analysis of each step:
The correct option is: [Ag(S₂O₃)₂]³⁻, Ag₂S₂O₃, Ag₂S
The formula Ag₂S₂O₃ for Y represents an unstable intermediate silver(I) thiosulfate compound that forms before the stable complex and is consistent with the sequence leading to hydrolysis.
This decomposition reaction is famously part of the "Iodine Clock" reaction. In one variation, arsenious acid (H₃AsO₃) reduces iodine to iodide, but the reaction is slow. However, if a small amount of sodium thiosulfate and starch are added, the iodine produced is immediately consumed by the thiosulfate (forming the complex X and Y). Only after all the thiosulfate is consumed and its complex hydrolyzes does the free iodine appear, reacting with starch to produce a sudden, dramatic blue color. The initial absence of color is due to the formation of the colorless silver-thiosulfate complexes discussed here.