Consider the following list of reagents:
Acidified K2Cr2O7, alkaline KMnO4, CuSO4, H2O2, Cl2, O3, FeCl3, HNO3 and Na2S2O3. The total number of reagents that can oxidise aqueous iodide to iodine is
K2Cr2O7 + H2SO4 + KI → K2SO4 + Cr2(SO4)3 + H2O + I2
KMnO4 + KOH + KI → KIO3 + K2MnO4 + H2O
H2O2 + KI → KOH + I2 + H2O
KI + Cl2 → KCl + I2
KI + O3 + H2O → KOH + I2
KI + HNO3 → KNO3 + H2O + I2 + [NO]
Na2S2O3 is acts as a reducing agent.
Iodide ions (I⁻) can be oxidized to iodine (I₂) by various oxidizing agents. The reaction is:
Any reagent with a higher reduction potential than iodine (E° for I₂/I⁻ is +0.54 V) can oxidize iodide to iodine. We will evaluate each reagent step by step.
Step 1: Acidified K₂Cr₂O₇ - It is a strong oxidizing agent (E° = +1.33 V). It oxidizes I⁻ to I₂.
Step 2: Alkaline KMnO₄ - In alkaline medium, it can oxidize I⁻ to I₂, though it might further oxidize to iodate in hot conditions. But generally, it does oxidize to I₂.
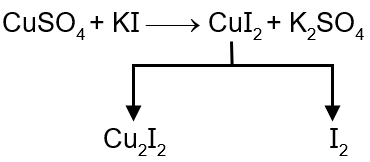
Step 3: CuSO₄ - Copper(II) ions oxidize I⁻ to I₂ (E° for Cu²⁺/Cu⁺ is +0.15 V, but reaction occurs due to low solubility of CuI).
Step 4: H₂O₂ - It oxidizes I⁻ to I₂ in acidic medium.
Step 5: Cl₂ - Chlorine is a stronger oxidizing agent (E° = +1.36 V) and oxidizes I⁻ to I₂.
Step 6: O₃ - Ozone is a powerful oxidizing agent and oxidizes I⁻ to I₂.
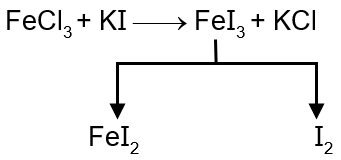
Step 7: FeCl₃ - Iron(III) oxidizes I⁻ to I₂ (E° for Fe³⁺/Fe²⁺ is +0.77 V).
Step 8: HNO₃ - Nitric acid oxidizes I⁻ to I₂.
Step 9: Na₂S₂O₃ - Sodium thiosulfate is not an oxidizing agent; it reduces I₂ to I⁻ (used in iodometric titrations). It does not oxidize iodide.
Final Answer: The reagents that can oxidize aqueous iodide to iodine are: Acidified K₂Cr₂O₇, alkaline KMnO₄, CuSO₄, H₂O₂, Cl₂, O₃, FeCl₃, and HNO₃. Na₂S₂O₃ cannot. Thus, the total number is 8.
The standard reduction potential (E°) determines the strength of oxidizing agents. An oxidizing agent with E° greater than +0.54 V can oxidize I⁻ to I₂.
General reaction:
Example with Fe³⁺: