For the reaction of type A(g) → 2B(g)
[Given [A] = concentration of A at tth time, [B] = concentration of B at tth time
a = initial concentration of [A] ]
| Column-I | Column-II |
| (A) If given reaction follow zero order kinetics then curve |
|
| (B) If given reaction follow first order kinetics then [B] Vs t curve |
|
| (C) If given reaction follow first order kinetics then half life Vs initial concentration curve | 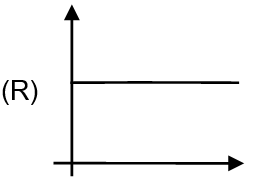 |
| (D) If given reaction follow zero order kinetics then half life(t1/2) Vs initial concentration curve |
|
 |
A —→ 2B
y = mX
A = S
Af = A0e–KT
Aused = A0 (1 – e–KT)
B = P
independent of time
C = R
A0 – Af = KT
D = ST
For the reaction: , we analyze the kinetics. Let [A] be the concentration of A at time t, [B] be the concentration of B at time t, and a be the initial concentration of A.
Since 1 mole of A produces 2 moles of B, the concentration of B at any time is given by: .
For zero order, (constant). From [B] = 2(a - [A]), differentiate: . So, is constant and directly proportional to . The graph is a straight line through origin with slope 1 (since if x-axis is and y-axis is , then y = 2x, but note the curve in column II: the first image shows a straight line through origin, which matches). So, (A) matches the first image.
For first order, . Then [B] = 2(a - [A]) = 2a(1 - e-kt). As t increases, [B] increases and approaches 2a asymptotically. The curve is exponential rise, similar to the second image in column II.
For first order reaction, half-life , which is independent of initial concentration. So, the graph is a horizontal straight line (constant). The third image in column II shows a horizontal line, so (C) matches.
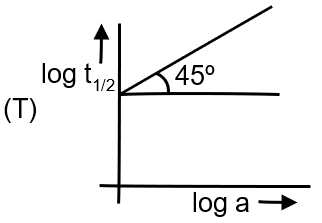
For zero order, . So, half-life is directly proportional to initial concentration a. The graph is a straight line through origin with positive slope. The fourth image in column II shows a straight line through origin, so (D) matches.
(A) matches the first curve, (B) matches the second curve, (C) matches the third curve, (D) matches the fourth curve.
Rate is constant: . Integrated rate law: . Half-life: .
Rate is proportional to concentration: . Integrated rate law: . Half-life: .
For reaction , the rates are related by .