For the reaction conc. at time t = 0,
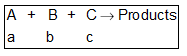
Let rate = kCACBCC2. Let b >> a and c >> a then rate is given by,
rate = k'CA. Choose the correct option.
No Solution
This problem involves a reaction with multiple reactants and a complex rate law. The key concept is simplifying the kinetics under specific concentration conditions to form a pseudo-first order reaction.
Step 1: Analyze the Given Rate Law
The rate law is given as:
Step 2: Apply the Given Conditions
The conditions state that the initial concentrations of reactants B and C are much greater than the concentration of reactant A ( and ). This means that as reactant A is consumed, the concentrations of B and C remain effectively constant throughout the reaction.
Step 3: Derive the Pseudo-First Order Rate Constant (k')
Since and are constant, they can be combined with the original rate constant to form a new, effective rate constant .
The rate law simplifies to:
Therefore, by comparing the two expressions, we find:
Since we are dealing with initial concentrations, we substitute with and with :
Final Answer: The correct option is .
1. Rate Law: An equation that shows the relationship between the reaction rate and the concentrations of reactants. For a general reaction , the rate law is , where and are the orders of the reaction with respect to A and B.
2. Reaction Order: The power to which a reactant's concentration is raised in the rate law. The overall order is the sum of all individual orders (e.g., ).
3. Pseudo-First Order Reaction: A higher-order reaction that is made to behave like a first-order reaction by keeping the concentrations of all but one reactant in large excess. The rate law simplifies to , where .
4. Integrated Rate Law for First-Order: For a reaction with rate law , the concentration of A at time is given by .