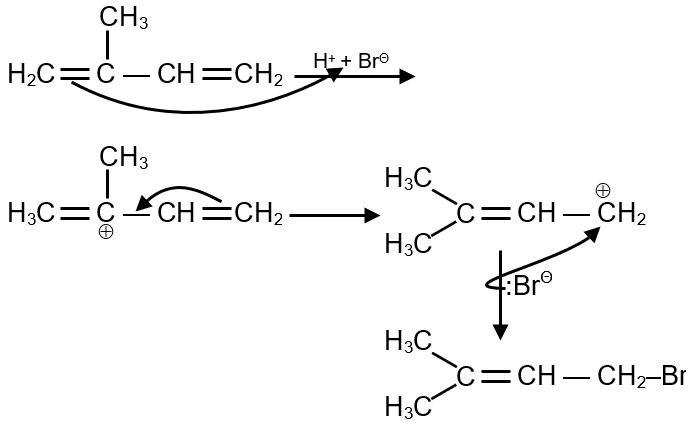
In the following reaction, the major product is:
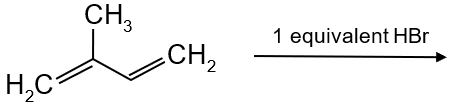
This question involves an electrophilic aromatic substitution reaction where an alkyl halide (CH3CH2Cl) reacts with benzene in the presence of anhydrous AlCl3 (a Lewis acid catalyst). This is known as Friedel-Crafts alkylation.
Step 1: Generation of Electrophile
The Lewis acid AlCl3 coordinates with the chlorine atom of CH3CH2Cl, polarizing the C-Cl bond and generating a carbocation electrophile.
Step 2: Electrophilic Attack
The electrophile (ethyl carbocation) attacks the electron-rich benzene ring, forming a sigma complex (arenium ion intermediate).
Step 3: Deprotonation
The arenium ion loses a proton (H+) to regenerate the aromaticity, yielding ethylbenzene as the final product.
Final Product: The major product is ethylbenzene , which corresponds to the first option.
Friedel-Crafts alkylation can sometimes lead to carbocation rearrangement, but with ethyl chloride (a primary halide), rearrangement is minimal under normal conditions. The ethyl carbocation is sufficiently stable for direct substitution without forming isopropylbenzene. The other options show different substitution patterns or rearranged products that are not favored here.
Friedel-Crafts Alkylation: A reaction where benzene is alkylated using an alkyl halide and AlCl3 catalyst to form an alkylbenzene.
Electrophilic Aromatic Substitution (EAS): A reaction where an electrophile replaces a hydrogen atom on an aromatic ring. General mechanism involves electrophile generation, attack, and deprotonation.
Carbocation Stability: The order of stability is tertiary > secondary > primary. Rearrangement can occur if a more stable carbocation can form.
In Friedel-Crafts alkylation, the electrophile is a carbocation. Primary carbocations may rearrange to more stable secondary or tertiary carbocations if possible, but with ethyl chloride, rearrangement to isopropyl is not significant under standard conditions, making ethylbenzene the major product.